Erysipelas – Parvovirosis – Leptospirosis
Control and prevention of these 3 pathogens are key to ensure good reproductive performance in breeding herds worldwide.

Leptospirosis
Disease
Leptospirosis is caused by a bacterium and is a zoonotic disease: an illness that can be transmitted from animals to humans. There are currently more than 200 serogroups and serovars identified. Pigs act as maintenance hosts for serovars Bratislava, Pomona and Tarassovi. Within a geographic region, certain serovars are prevalent and become adapted to a particular maintenance host, while only the serovar Bratislava has worldwide distribution.
Most important leptospiras reported in pigs:

Source: García Peña F. J., Fraile L. Leptospira 1. Introduction and pathogenesis. www.msd-animal-health.com. First published in International Pig Topics volume 31.1
Although there are no exact references of the prevalence of the disease, evidence indicates it is abundant worldwide. The prevalence of infection with the different serovars also varies between countries, depending on the presence of maintenance hosts and of their contact with the pig population. The rates also vary depending on the laboratory technique used to diagnose the disease. In Spain it is estimated that approximately 85% of the farms tested at a national level are positive to some pathogenic Leptospira serovar.
Leptospirosis affects the reproductive system of gilts and sows. When a herd becomes infected, the disease spreads continuously. The infected sows become carriers. Although it is true that the bacteria have very effective means of inmune evasion. The maternal immunity does not last long after weaning.

Economic impact
When leptospirosis affects a herd of susceptible sows for the first time, it causes:
Abortions and stillborn piglets: Surge in abortions together with an increase in the number of stillborn piglets.
Weak piglets: Piglets that are born weak.
Other effects: Is endemic leptospirosis (chronic, subclinical) that causes the greatest economic losses due to its continuous and hidden effects.
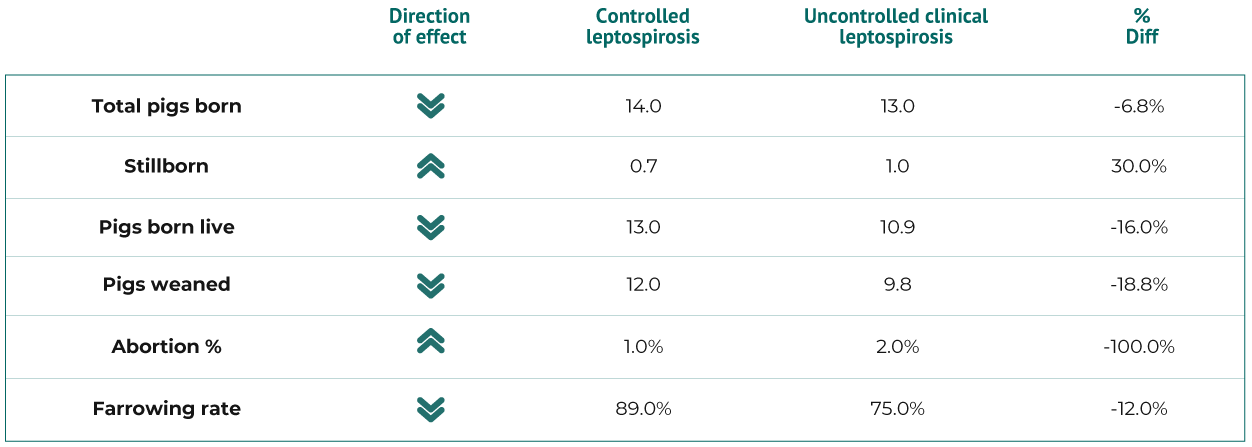
According to a study1 performed in the USA, a 5,000-sow farm affected by clinical leptospirosis produces 30,000 weaned piglets less per year than a farm without clinical leptospirosis.
According to this study, the farm with controlled leptospirosis spends USD29.07 for producing a piglet, whilst a farm with clinical leptospirosis spends USD37.35 per piglet. It estimates that the expenses of a 5,000-sow farm with or without leptospirosis are almost the same, but the one with controlled leptospirosis weans 30,000 piglets more, therefore reducing the total costs.
In UK, Leptospira outbreak lasting four months led to a 300-sow herd suffering a drop in annual production of 1.3 pigs per sow per year or 400 pigs less weaned. At a marginal cost of £35 per pig, this implied a cost of £14,000.2
The costs of non-controlled Leptospira could reach up to $8.28 per piglet.1
Sow Herd Productivity Effects of Uncontrolled Clinical Leptospirosis1

Prevalence
Seroprevalence of the disease varies considerably between countries and even between different regions in the same country. Actinobacillus suis can cause similar disease and lesions but is not as contagious as Actinobacillus pleuropneumoniae.
Abundant Worldwide
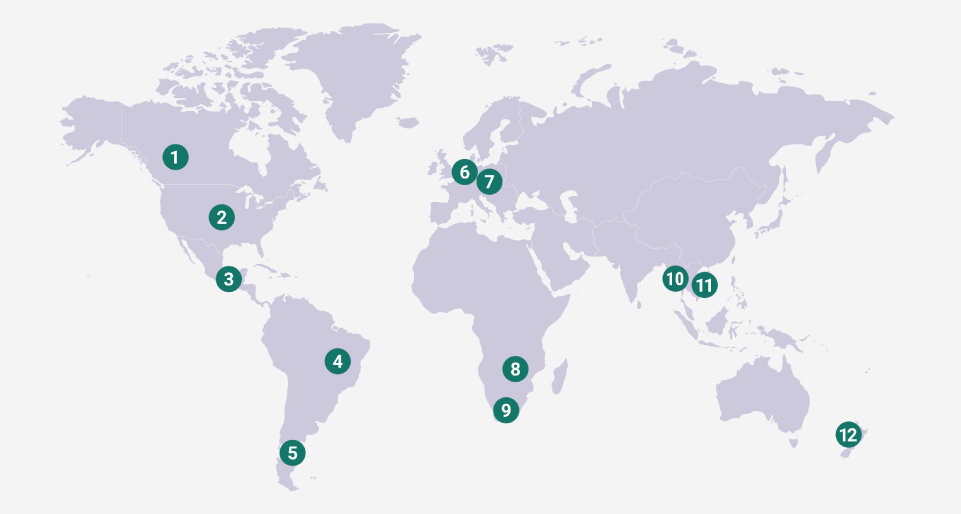
SEROPREVALENCE OF THE DISEASE VARIES
Seroprevalence of the disease varies considerably between countries and even between different regions in the same country.

Based on: Garcia Pena, Francisco Javier, Essential guides on swine health and production, leptospirosis, Servet 2018.
1.Prevalence on a farm level: animals from different farms. Were tested and farms that had at least one positive animal were considered positive.
2.Individual prevalence: percentage positive out of all the animals tested, regardless of the farm of origin.
3.Prevalence of infected animals: percentage positive for a particular serve out of all animals infected by leptospira.
An overall Leptospira seropositivity of 20.2% has been recently reported in Germany, with an increasing trend over time, between 16.3% in 2011 and 30.9% in 2016.3

Diagnosis
Pigs infected with Leptospira rarely develop clinical signs themselves, they often go unnoticed in mature, nonpregnant swine or in growing pigs. Reproductive failure, as evidenced by infertility and sporadic abortion in late gestation, is the most common clinical sign in gestating sows, and venereal transmission may occur. In many cases, foetuses are carried almost full term but may be mummified, dead, or weak at birth.
Finishing pigs may have scattered foci of interstitial nephritis or generalized kidney scarring which may only be noticed at slaughter as “white-spotted kidneys”.
Key points to recognize leptospirosis:
- Abortions, particularly in late gestation.
- Increase in weak pigs born, stillbirths and mummification.
- Infertility (regular and irregular returns to oestrus during first weeks of pregnancy).
Laboratory tests:
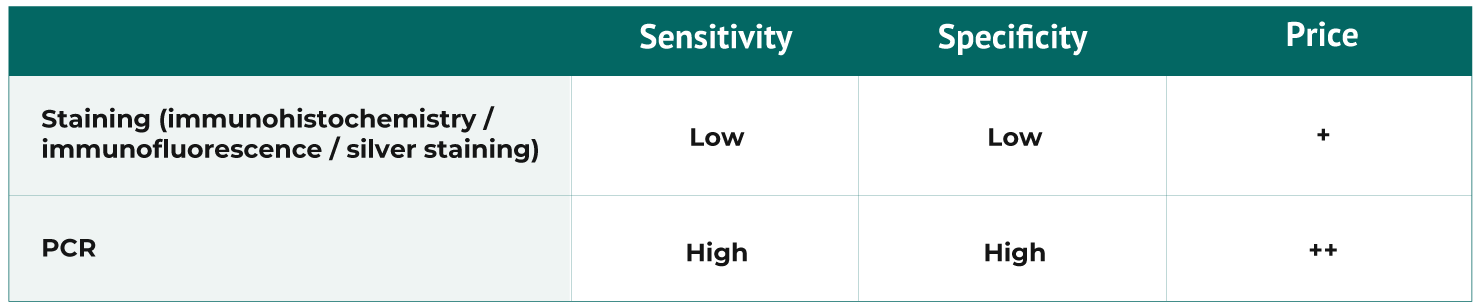

Serology (MAT) is valuable under the following circumstances:
- To identify seroconversion in a sow with an acute infection and symptoms we recommend leaving 14-21 days between pre-and-post-serum.
- To identify a leptospirosis infection during gestation by testing the thoracic fluid of aborted foetuses (gestation > 80 days) or for newborn piglets before colostrum intake.
Antibodies can be detected from 5-10 days after infection to no more than approx. 3 weeks after infection.
When taking blood samples after farrowing (result of stillborn piglets and lots of mummies), it may be too late to prove seroconversion. Afterward the antibody titre will drop relatively quickly. Many blood samples are required to map out an endemic leptospirosis infection.
For the serovars for which pigs are natural hosts (Pomona, Tarrasovi, Bratislava), the antibody titers are often low. No distinction can be made between antibodies after infection or after vaccination.

Treatment and prevention
Prevention solutions should be prioritized to limit the use of antibiotics and therefore, the risks of antibiotic resistance.
Control measures should include strict biosecurity, hygiene protocols, vaccination and medication when needed.
- Biosecurity, including strict rodent and bird control programs to minimize infection from other animal species, as rodents can be carriers of Leptospira.
- Hygiene, including cleaning and disinfection protocols of all buildings/rooms between batches of pigs. Leptospira is sensitive to several detergents and disinfectants, so a good cleaning protocol will be basic.
- Vaccination, to improve immunity and reduce disease prevalence. It helps protect the animals’ health, reproductive performance and farm productivity. Once the vaccine has been introduced, it is recommended to use it regularly in all the sows and gilts on the farm, therefore minimizing the possibility of transmission.
- Medication, if a leptospirosis outbreak occurs. Antimicrobials are usually effective controlling clinical signs, although they do not eliminate infection completely.
Medication, Leptospirosis is likely to remain endemic in swine herds, however eradication programs have been reported. Eradication of Leptospira has been performed in swine populations raised under high, strict biosecurity conditions using a combination of medication, with or without vaccination, together with measures to eliminate environmental infection.
Leptospirosis is a contagious disease of swine and many other animal species, including humans. Therefore, it is a potential occupational zoonosis with public health implications.
1 Stein T. https://www.leptospirosis-in-swine.com/economic-impact
2 White M. Leptospirosis in pigs. NADIS. 2016
3 Strutzberg-Minder K, Tschentscher A, Beyerbach M, Homuth M, Kreienbrock L. Passive surveillance of Leptospira infection in swine in Germany. Porcine Health Manag. 2018.
Download the disease infographic
MSD Animal Health solutions to control Erysipelas – Parvovirosis – Leptospirosis

SowCare: Porcilis® Ery+Parvo+Lepto
Visit https://www.leptospirosis-in-swine.com/ for more information.
Porcilis® Ery+Parvo+Lepto is an inactivated vaccine against Porcine parvovirus, Erysipelas disease and Leptospira.
SOLUTIONS
Porcilis® Ery+Parvo+Lepto
Inactivated vaccine against Porcine Parvovirus,
Erysipelas disease and Leptospira.


Onset of immunity after vaccination:
E. rhusiopathiae: 3 weeks
Porcine Parvovirus: 10 weeks
Leptospira serogroups: 2 weeks
Duration of immunity:
E. rhusiopathiae: 6 months
Porcine Parvovirus: 12 months
Leptospira serogroups Australis: 2 weeks
Leptospira serogroups Canicola, Icterohaemorrhagiae, Grippotyphosa, Pomona and Tarassovi: 12 months
BENEFITS
- The broadest protection against Leptospira.a
- The longest combined protection for the 3 pathogens.
- Excellent safety profile.b
- Can be used during pregnancy.c
HOW TO USE IT
- For intramuscular use.
- Administer a single dose of 2 ml in the neck region.
Available in EU, APSA and LATAM
Porcilis® Ery+Parvo+Lepto can be used in mass vaccination.
a. Jacobs, A.A.C., et al. (2015). A novel octavalent combined erysipelas, parvo and Leptospira vaccine provides (cross) protection against infection following challenge of pigs with 9 different Leptospira interrogans serovars. Porcine Health Management 1:16 pp 1-7.
b. Jacobs, A.A.C., et al. (2015). Safety and efficacy of a new octavalent combined erysipelas, parvo and Leptospira vaccine in gilts against L. interrogans serovar Pomona associated disease and foetal death. Vaccine 33 pp 3963-3969.
c. Porcilis® Ery+Parvo+Lepto SPC

SowCare:
Other related vaccines
Porcilis® Ery+Parvo
Porcilis® Ery
Porcilis® Parvo



