
APP – Actinobacillus pleuropneumoniae
Disease
Actinobacillus pleuropneumoniae (APP) is one of the most important swine bacterial respiratory pathogens found worldwide. This bacterium is the etiologic agent of porcine pleuropneumonia, which is a highly contagious respiratory disease.
APP has at least 18 different serotypes, some of which do not produce any clinical signs, while others may cause serious disease. The serotypes vary depending on the country. Serotypes 1,2,5,9 and 11 are normally quite virulent (2 and 9 are highly virulent in many European countries), while serotypes 3 and 7 are quite mild.
All APP strains, even low virulent ones, can also act as secondary pathogens contributing to increased mortality in conjunction with other viral and bacterial pathogens as a component of the porcine respiratory disease complex (PRDC).
APP persists in the tonsils and the upper respiratory tract, infected animals may not show any clinical signs. This is the main mechanism for the introduction into herds. It is transmitted over short distances through aerosol and only survives outside the pig for a few days.
Although chronic cases may appear following acute outbreaks, the disease is often characterized by sudden onset, peracute or acute clinical course, and high morbidity and mortality.
A. pleuropneumoniae causes respiratory disease that affects the lungs:
In acute outbreaks, affected pigs can exhibit breathing difficulties and depression. Lungs become hard and haemorrhagic, excess fluid gathers in the chest cavity, and pigs can die suddenly.
In chronic infections, daily weight gain and feed conversion rate is reduced, leading to long-term economic loss.

Economic impact
The daily weight gain and the feed conversion rate of infected pigs is reduced, while mortality as well as medication and veterinary expenses are increased.
Annual costs of APP, depending on the severity of the disease, have been estimated to reach up to $27 (€24) per pig space for farms with low bio-security.1
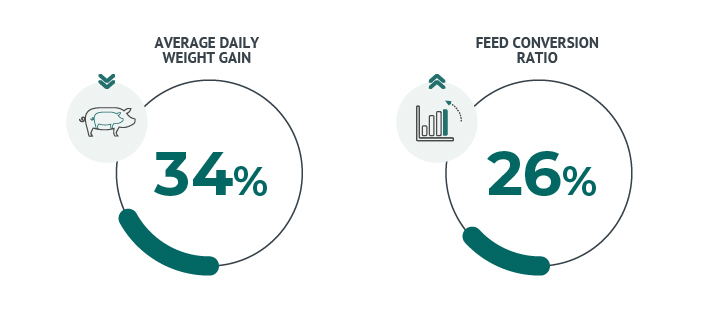
The economic impact is not limited to the mortality rate (which can be as high as 10%) but also treatment costs as well. Average Daily Profit can be reduced by up to 34% while the deterioration of the feed conversion rate can reach 26%.2

Prevalence
APP is widely distributed throughout major swine-raising countries. It has been a major problem in many European countries and is relatively common in the United States, Canada and Asia.
In naïve herds, APP occurs in all age groups but usually is seen in 6-20-week-old pigs.3
Actinobacillus suis can cause similar disease and lesions but is not as contagious as Actinobacillus pleuropneumoniae.
In the US, it is claimed that 80% of farms are infected but disease is only seen in 20%.4
In a Spanish report, almost 90% of studied herds were serologically positive for APP.5
In a Canadian study, 78% of herds were APP positive based on PCR detection in the upper respiratory tract of piglets.6

Diagnosis
Based on the farm history, clinical signs, postmortem examination including slaughterhouse exams and culture of the organism. Pulmonary lesions are very typical (lung monitoring at slaughter can help diagnosing chronic cases). However, diagnosis is confirmed by culture, identification, and often serotyping of APP. Serology may be used to identify serotypes, but its interpretation can be difficult due to cross reactivity between serotypes.
APP must be differentiated from Actinobacillus suis infections, enzootic pneumonia, PRRS, influenza, and pneumonia caused by Salmonella choleraesuis.
Clinical signs depend on whether it is an acute/peracute outbreak or chronic infection.
Acute or peracute outbreak:
- Severe breathing difficulties often evidenced by mouth- and abdominal breathing. Pigs sit down to make breathing easier.
- Coughing.
- Haemorrhagic foam discharged from the nose or mouth just prior to death.
- High fever.
- Anorexia, depression, and refusal to move.
- Cyanosis (blueing of the ears, nose, legs, and abdomen).
- Sudden death.
Chronic infection:
- Spontaneous or intermittent coughing.
- Slightly increased mortality.
- More pleurisy discovered at slaughter.
- Low-level, long-term losses.
Serology:
ELISA can be used to perform routine surveillance of possibly infected herds. Due to the different types of test available, the possibility of cross-reactions between serotypes, and the virulence differences between APP strains, a careful interpretation is needed, especially in the absence of clinical disease.
There are several tests available for routine diagnosis of APP:
- ELISA.
- PCR.
- Microbiological culture.

Treatment and prevention
Acute outbreaks should be treated with injectable antibiotics to reduce mortality, whilst chronic APP needs an effective vaccination program. If the outbreak occurs in piglets up to 10 weeks of age, consider sow vaccination.
If APP is endemic in a sow farm, depopulation can be an adequate approach to eradication of the disease. Depopulation should be followed by thorough cleaning and disinfection of the buildings and leaving them empty for a few weeks; restocking can then follow using APP-free pigs.7
In all cases, farm management should be reviewed to minimize the spread of infections. Farms free of APP should only introduce stock from APP-free herds and new stock should be quarantined.
1 Stygar A.H. et al, Economic value of vaccination against Actinobacillus pleuropneumoniae in a fattening pig herd. Conference NJF seminar 476. Economics of Animal Health and Welfare. 2014.
2 Straw B., 1989.
3 Actinobacillus pleuropneumoniae. Faculty of Veterinary Medicine, Vietnam National Univ. of Agriculture. 2020.
4 Mark White. NADIS (The National Animal Disease Information Service). www.nadis.org.uk. 2007.
5 Fraile et al. 2010.
6 MacInnes JI et al. Can J Vet Res. 2008.
7 Sassu EL, Bossé JT, Tobias TJ, Gottschalk M, Langford PR, Hennig-Pauka I. Update on Actinobacillus pleuropneumoniae-knowledge, gaps and challenges. Transbound Emerg. Dis. 2018.
Download the disease infographic
MSD Animal Health solutions to control APP – Actinobacillus pleuropneumoniae

ResPig: Porcilis® APP
ResPig Porcilis® APP protects pigs against infection with Actinobacillus pleuropneumoniae. It is a multivalent toxoid vaccine that protects against all known serotypes of APP.
SOLUTIONS
Porcilis® APP
Active immunization of weaning pigs as an aid in the control of pleuropneumonia caused by Actinobacillus pleuropneumoniae.


Onset of immunity: 2 weeks after completion of the vaccination scheme
Duration of immunity: 11 weeks after completion of the vaccination scheme
BENEFITS
- No need for serotyping.
- Reduced mortality and clinical signs.
- Reduced lesions of pleuropneumonia caused by APP.
HOW TO USE IT
- Two doses of 2ml, from 6 weeks of age with a 4-week interval between doses.



